Sirona Dx is the first CRO to provide access to the latest spatial multiomics 3D imaging technology from Nanostring.

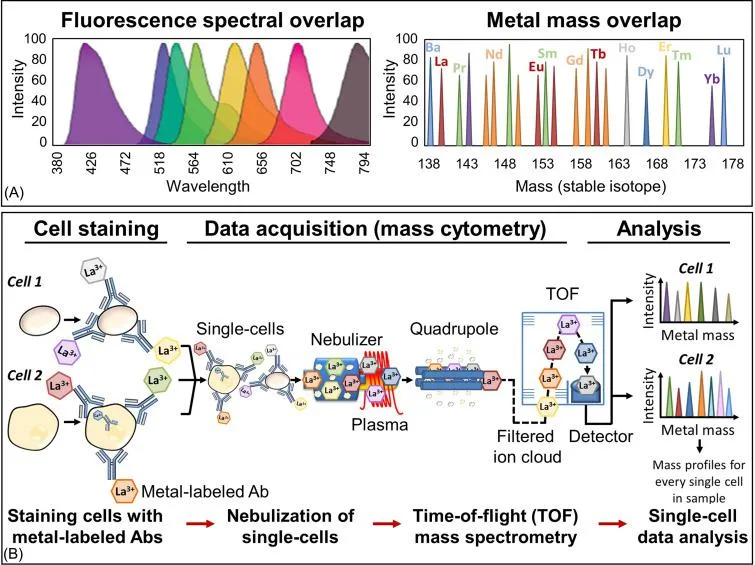
Comparison of fluorescence and mass spectra overlap and typical schema of sample preparation and analysis of single cells by mass cytometry. (A) Histograms demonstrating the emission spectra overlap that exists between different fluorophores within a fluorescence-labeled flow cytometry antibody panel compared to the metal mass overlap that exists for heavy metals within a mass cytometry metal-labeled antibody panel. (B) Sample preparation and analysis by mass cytometry schema. Cells are stained with a metal-labeled antibody cocktail targeting specific antigens of interest under resting or stimulating conditions. Cells are fixed prior to mass cytometry acquisition to preserve the cell state/antibody binding and washed to remove salts and unbound antibodies. Upon entering the instrument, cells travel through an argon plasma that vaporizes and ionizes the cells, antibodies, and the attached heavy metal tags. Each cell is converted into an ion cloud and passes through a quadrupole filter, which removes all ion masses below approximately 75 Da. The remaining heavy-metal reporter ions in the ion cloud are separated by their mass-to-charge ratio as they accelerate towards a detector in a time-of-flight mass spectrometer. The time-resolved detector measures a mass spectrum that represents the identity and quantity of each isotopic probe on a single-cell basis.
From: Alan D. Michelson, Platelets (Fourth Edition), Academic Press, 2019.

